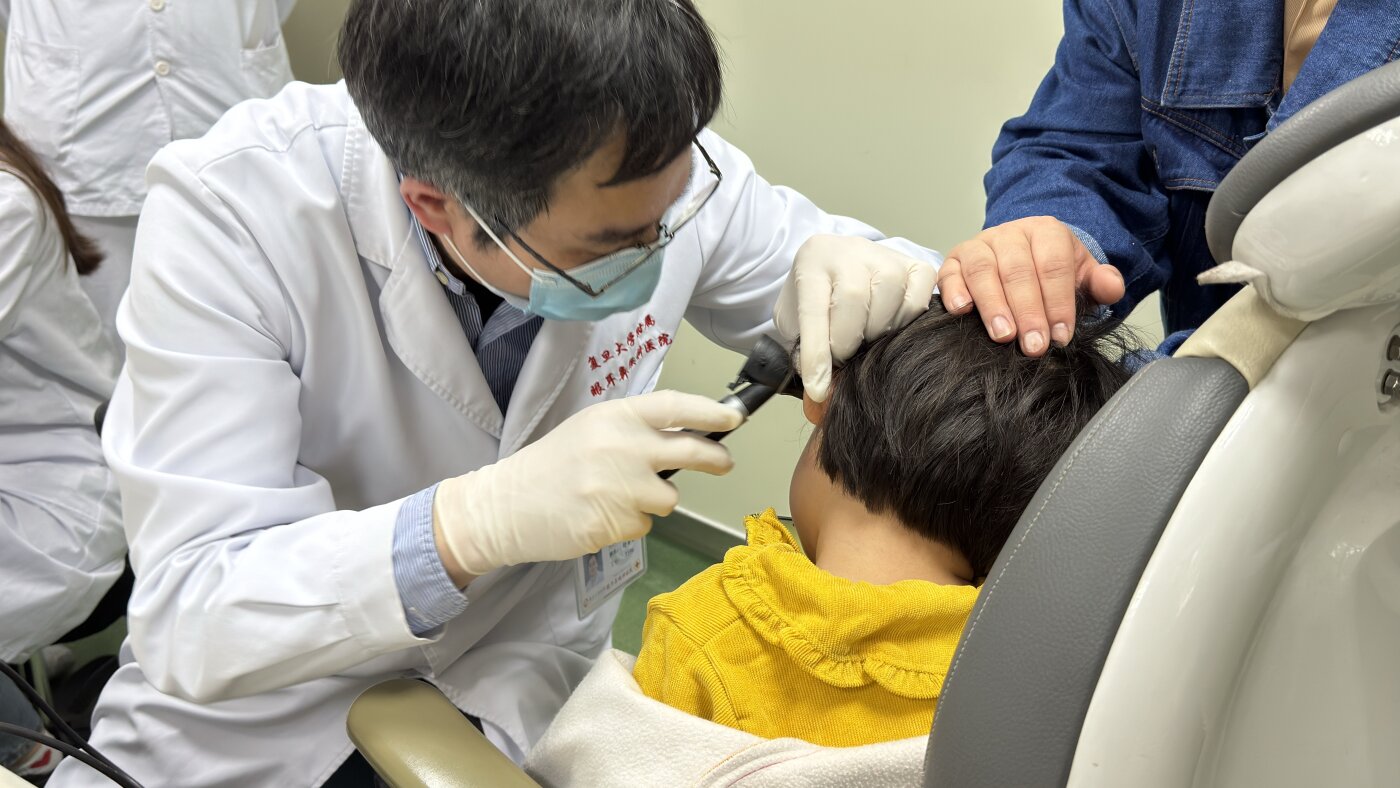
FDA approves first gene therapy for genetic hearing loss
The Food and Drug Administration on Thursday approved Regeneron’s Otarmeni, the first gene therapy for genetic hearing loss. Subscribe to read this story ad-free Get unlimited access to ad-free articles and exclusive content. The drugmaker said it will offer the drug for free to U.S. patients. Otarmeni is approved to treat a very rare form of hearing loss that affects about 50 babies born in the U.S. each year and is caused by a mutation in a gene called OTOF. The approval was granted under the Commissioner’s National Priority Voucher, an FDA pilot program intended to fast-track drug reviews. “This really is life-changing for families with children with hearing loss,” Dr. Eliot Shearer, a pediatric otolaryngologist at Boston Children’s Hospital and a principal investigator on the Regeneron trial. The only other treatment option for children with genetic deafness is cochlear implants, Shearer said, which restore the ability to hear speech and music, but reduce the fidelity of the sound. With gene therapy, the improved hearing is “on 24/7 and doesn’t rely on batteries,” he added. …